Derivation of the Rate Law for the the Reversible Michaelis-Menten Mechanism
http://biology.stackexchange.com/a/43832/1136
Created with Wolfram Mathematica
1. Preliminaries
![]()
![]()
2. Mechanism
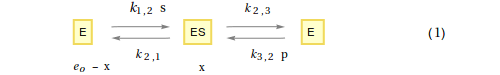
3. Derivation of the rate-constant form of the Rate Law
Set up the differential equation
Let $x$ be the conentration of ES
Let $e_o$ be the total enzyme concentration
Therefore, the concentration of E (the 'free' enzyme concentration) equals $e_o$ - $x$
From the steady-state assumption, the rate of formation of x will equal the rate of breakdown of $x$, and the following differential equation may be written
$$ {dx\over dt} = {k_{1,2}\ (e_o -x)\ s + k_{3,2}\ (e_o -x)\ p - (k_{2,1} + k_{2,3})\ x = 0}\ \ \ \ \ \ \ (2)$$
Solving for $x$
![]()
![]()
4. Velocity equation
![]()
![]()
![]()
![]()
5. Define Kinetic Constants
![]()
![]()
![]()
![]()
6. The Kinetic-Constant form of the Rate Law
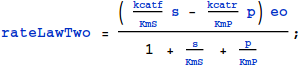
7. An Important Check
![]()
![]()